
Calcium bromide
The Importance of Calcium Bromide in Drilling
Drilling operations rely on advanced technologies and chemical solutions to overcome challenges. Calcium bromide and drilling starch are two vital components that ensure stability and efficiency in the process. For effective use, selecting the right chemicals for specific purposes is crucial. Calcium bromide, along with zinc and sodium bromides, addresses high temperature and pressure challenges in drilling oil and natural gas wells. Proper chemical choices help reduce costs and improve project outcomes.
Drilling Fluids and Their Role
Drilling fluids play a critical role in maintaining well stability. They regulate pressure, remove cuttings, and cool drill bits during operations. These fluids also prevent premature well closures by reinforcing well walls. The use of anti-corrosion additives, like iron oxide, prevents chemical reactions that can damage oil field equipment. Combining drilling starch with calcium bromide enhances the efficiency of drilling fluids by improving absorption and fluid loss prevention.
Importance of Calcium Bromide Solution
The stability of crude oil prices has increased investments in oil and gas projects, creating demand for reliable solutions like calcium bromide. This chemical, used in clear brine fluids, protects reservoirs and reduces mercury emissions from coal-fired power plants. At high combustion temperatures, it effectively absorbs elemental mercury released during coal burning.
Where is it Found?
Calcium bromide (CaBr₂) is not naturally occurring and is synthesized through chemical reactions. Its production involves calcium carbonate (CaCO₃), calcium oxide, and hydrobromic acid (HBr). The compound’s molar mass is 199.89 g/mol, and it is widely used due to its stability and efficiency in industrial applications.
Chemical Formula
A 52% calcium bromide solution is a dense, clear brine fluid with a specific gravity of 1.7 kg/L. This formulation ensures consistent performance in demanding environments.
CAS Number: 7789-41-5
Uses: Workover fluids, mercury emission control, and drilling fluids
Calcium Bromide Solution 52%
Calcium bromide 52% solution serves multiple purposes in oil and gas operations. It is used as a clear brine fluid or mixed with calcium chloride or zinc bromide to achieve desired densities and crystallization points. Its primary use is as a completion and workover fluid, controlling wellbore pressures in upstream drilling activities.
Other Uses of Calcium Bromide
Beyond drilling, calcium bromide finds applications in various industries:
Photography: For developing solutions
Medicine: In specific pharmaceutical formulations
Wood Preservation: As a preservative
Flame Retardants: In protective blends
Sizing Compounds: For textile and paper production
Is Calcium Bromide Soluble in Water?
Calcium bromide dissolves easily in water, methanol, ethanol, and acetone. However, it remains insoluble in organic solvents. Its melting point is 730°C, boiling point is 1935°C, and density is 3.35 g/L, making it ideal for high-temperature drilling applications.
Why Choose Calcium Bromide and Drilling Starch?
Together, calcium bromide and drilling starch provide reliable and cost-effective solutions for oil and gas drilling. Their combined benefits ensure well stability, corrosion prevention, and efficient fluid performance.
MSDS of Calcium Bromide!
Calcium bromide Technical Specification
Properties | Value |
Molecular Weight Table of Contents Toggle | 199.90 |
Cas No. | 7789-41-5 |
Appearance | Transparent Water White Liquid |
Purity | 52% min |
Physical State | Heavy Density Liquid |
Assay | 52%by weight CaBr2 |
Bulk Density | 14.2 Ib/gal |
Solubility | Completely Soluble |
PH Value | 7-10 |
Mol Formula | CaBr2 |
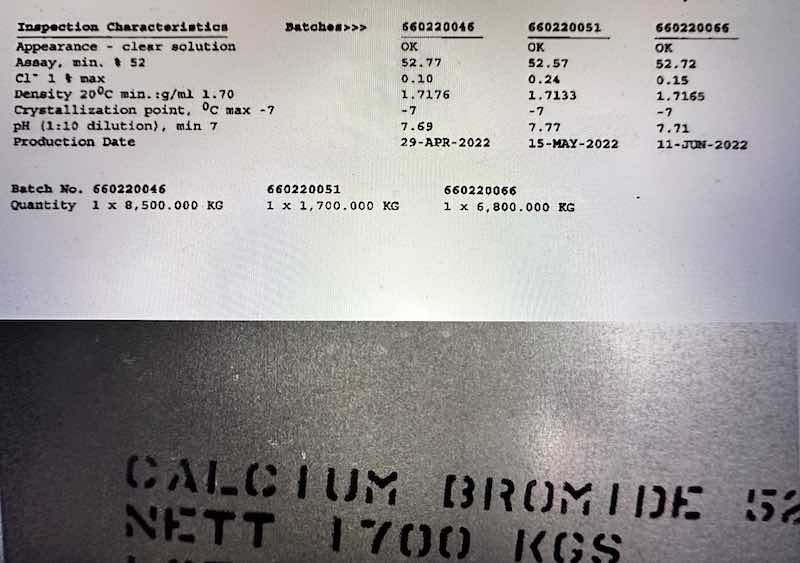
Table of Contents
Share this Content
Facebook
Twitter
LinkedIn
Contact Info
UAE: No. 3509 of Burlington Tower, Business bay, Dubai
Turkey: No. 6, 613 Sk., Galip Erdem Cd.,İlkbahar Mah, Cankaya, Ankara
Maslak Mahallesi, Söğüt Sokak No: 20 T4A Blok, Daire: 219 Sarıyer – İstanbul, Türkiye
DIRECTORY
Recent News
CMC VS PAC: Complete Comparison Analysis
30 Apr 2026- Soda Ash in Glass Manufacturing: A Complete Guide08 Apr 2026
About Us
(ATDM) is Gilsonite miner located in Hakkari near to Iraq boarder.
